CBD Labeling Guidelines and Requirements
- Jun 6, 2019
Every so often a new miracle health product takes the market by storm. Years ago, quinoa was all the rage. At another point in time, acai set the world ablaze. It seems, though, that in this latest iteration of the trend we find a product with a greater range of applicability than any of its predecessors. That product, which you are probably aware of from the title, is CBD.
Your CBD product may come with some marked health benefits, but in order to get that product in front of customers, you’ll need to make sure your label is in keeping with CBD labeling guidelines. Below we will give you some tips on how to ensure your cannabis labels are compliant.
CBD labeling regulations are not prescribed by the federal government so, at times, they can be difficult to navigate. Check out the article below for a better understanding of CBD labeling guidelines.
“Note: This article is not intended to be legal advice. Nor is it meant to encapsulate the entirety of CBD labeling restrictions, especially where individual state mandates are concerned. It is meant to present an overview of CBD labeling regulations and their importance. It offers certain considerations that may benefit the designer of a CBD label should she or he hold them in mind.”
Classifying Your CBD Product
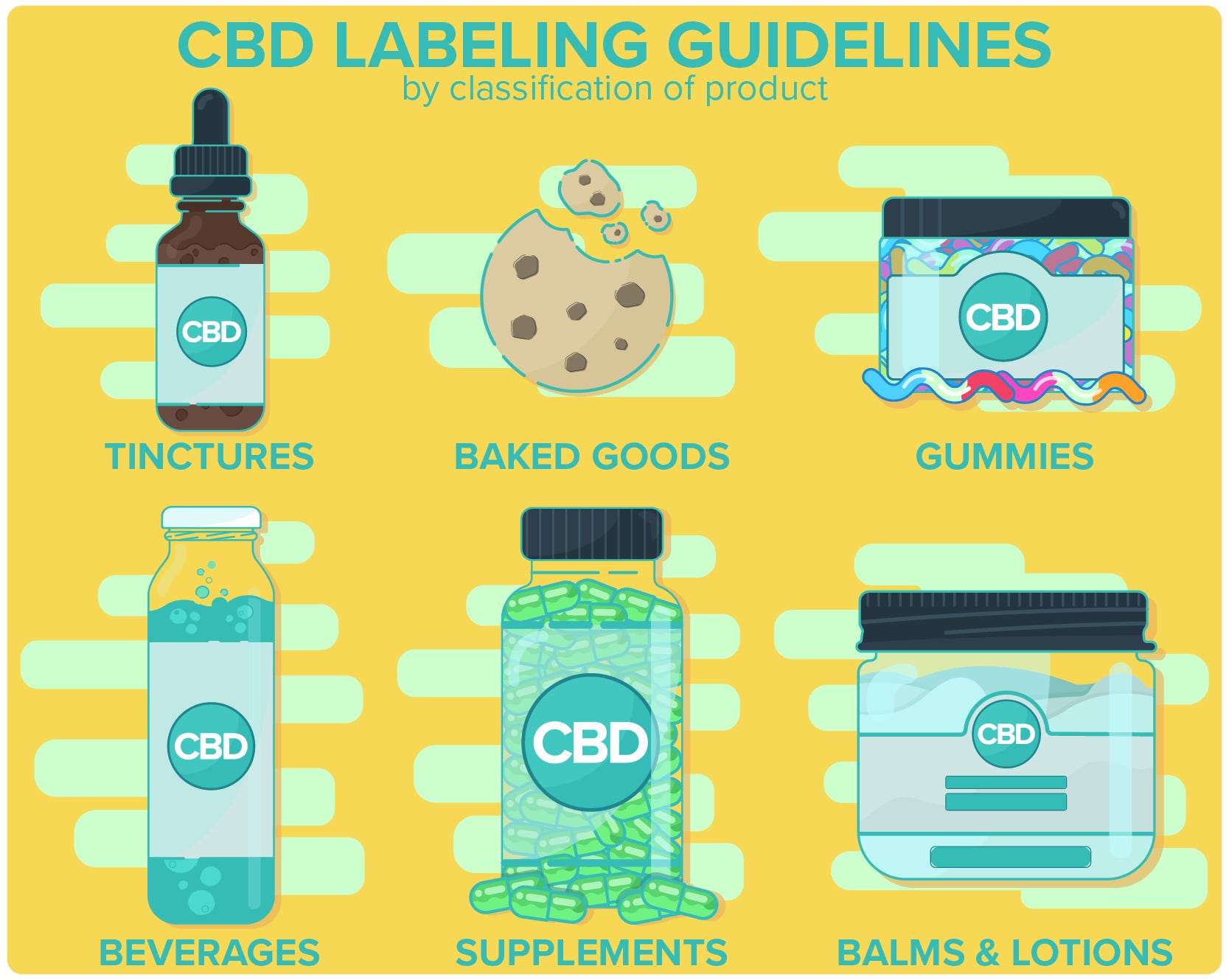
Firstly, you’ll need to determine whether your product qualifies as food or a supplement. Foods and beverages containing CBD are subject to different CBD labeling regulations than the ones set for CBD isolates. If the product qualifies as a food or drink, it will be subjected to the stipulations and regulations outlined by the FDA Food Labeling Guide. If it is a supplement then you’ll need to consult the FDA labeling regulations for dietary supplements.
If the suggested serving size for a food or beverage is below the FDA’s RACC (recommended amount customarily consumed), then the formatting should be consistent with that of a supplement.
Many, if not most, hemp-CBD products can be classified as supplements. Keep in mind that no matter what you classify your product as, CBD labeling regulations say that you must clearly articulate what the product contains.
What’s In Your CBD Product?

If you’re unsure, or simply want to support claims, about the makeup of your CBD product, consider getting a certificate of analysis. A certificate of analysis, or COA, is granted to those companies that allow their products to be analyzed by a third party. Doing so, will allow you to more accurately list the percentage of CBD, and each of the other ingredients, in your product.
Many sellers of CBD have no clue as to what is in their products. Need proof? Ryan Vandrey, an Associate Professor of Psychiatry and Behavioral Sciences at John Hopkins School of Medicine, published a 2017 study in Jama that gives insight into the issue. After analyzing 84 samples of CBD tinctures, vaporization liquids and oils from 31 different companies, he found that many had CBD content incongruent with that stated on their packaging.
Twenty-six of the 84 products had CBD levels higher than what was stated on their labels. High enough, in fact, to intoxicate the adults and children that needed them. Eighteen of those 84 samples had CBD levels far lower than that stated on their labels. In case you haven’t been doing the math, that’s a little over 50%. For consumers, and brand owners concerned about liability, that presents quite the gamble.
A more expansive study, conducted by analytic chemistry lab Ellipses Analytics, found similarly disturbing results. This study involved thousands of CBD samples and found that 70% of CBD products were “highly contaminated” with some amount of lead, arsenic, herbicides, BPA or toxic mold.
One company that shall remain nameless (pssst, it was Ananda Hemp) had CBD with levels of lead 100 times higher the EPA’s recommended limit for drinking water. To be fair, the company did argue that the findings were false.
The point is that brand owners should be wary of what is in their products. Consumers, for their sake and yours, should be privy to the list of ingredients included in your CBD product. That said, let’s delve further into the CBD labeling guidelines to find out how to list the ingredients in your product.
CBD Labeling Regulations Differ Depending on the State

The specific CBD labeling regulations pertinent to the making of your label design may depend greatly on the state you’re operating out of. States with medical and recreational cannabis laws, understandably, have stricter laws regarding the presentation of the label. There are states, for example, that demand that the specific amount of THC and CBD be listed on the product’s label, even if that amount is 0%. Let’s take a look at the California’s policy to make matters more concrete. CBD labeling regulations state that “if the cannabis product contains less than 2mg of the cannabinoid and labeled prior to testing, the content may be listed as ‘
How to List Ingredients on Your CBD Label
One of the main things you’ll want make clear is whether or not the product you’re presenting is a CBD isolate, or simply contains CBD. Again, if it is a food or beverage item, the rear label should be designed in accordance with the FDA Food Labeling Guide. The identity of the product should be articulated on the front of the label. It should be written as “hemp extract supplement,” “herbal supplement,” or “dietary supplement,” depending on what the product is. Render the amount of a specific nutrient such as the total hemp extract or CBD oil in milligrams per serving. This is usually rendered in forms such as “250mg, 500mg, 750mg, or 1000 mg.” If the product is a supplement, then the rear of the package demands you include a Supplement Facts panel. This panel will be aesthetically similar to the Nutrition Facts panel, typically reserved for food items.
Via: Hemp Bombs
If your product contains any carrier oils, extra herbs, artificial coloring, sweeteners or flavors include those as well. CBD vape products will typically contain Propylene Glycol (PG) or Vegetable Glycerine (VG). Also while, an ingredient may not be active, it should still be included in the ingredients list.
If it is considered an herbal product, those products not considered foods, it is required that you state the specific parts of the plant used for the product. If it is a CBD isolate, then include a facts panel and render it as CBD per serving. You can also articulate each kind of cannabinoid included and list the amount of each per serving. These should be displayed as milligrams as well.
Including Serving Size on Your CBD Label
Serving size helps to alert the user to the amount of CBD in the bottle. It also allows them to get an accurate picture of the size of the dropper. Droppers will typically range in size from 0.5 millimeters to 1 millimeter. Maybe you have a hemp-extract product. Your hemp-extract product may be a direct CBD substitute, or it may simply possess cannabinoids content. If it is a direct substitute then there will be a direct 1:1 correlation. For example, 1000mg hemp = 1000mg CBD. If you are articulating the total cannabinoid content in your product, then the CBD will be less than the total sum of the product’s parts. So 1000mg of hemp extract may be the equivalent of 700mg of CBD.
Via: Hemp Bombs
Benefits of Following CBD Labeling Guidelines
The FDA is not hesitant about taking companies to task for making products that are in clear violation of CBD labeling guidelines. The making of unsubstantiated claims about the benefits of CBD products are, of course, strictly prohibited. That, however, hasn't stopped companies from trying to promote such claims. Earlier this year, they sent out what we can only assume were sternly-worded emails to NutraPot, PotNetwork Holdings and Advanced Spine and Pain. The companies were chided for the claims included on their label. Most notably, the idea that CBD could be used to alleviate the troubles of cancer and Alzheimer’s disease. Claims, essentially, with no scientific evidence to back them.
So maybe CBD is not the panacea it is touted to be. That’s fine. Like Acai, it has health benefits that do approach the miraculous. Harvard Medical School reports that CBD is effective in treating many debilitating diseases. Epilepsy syndromes, like Dravet syndrome and Lennox-Gastaut syndrome (LGS), for example.
https://youtu.be/iWF9MmiQUA4
Not only does the scientific literature back these claims, but it testifies to CBD’s effectiveness as a treatment for anxiety disorders, insomnia, chronic pain, arthritis, and neuropathic pain. The availability of a product like CBD must seem like a godsend for those afflicted with these ailments. It is important then, that brand owners maintain a certain level of integrity and consistency when presenting their products and designing their hemp labels. Put simply, consumers should know what they’re getting.
Thanks for reading and hey, if you’d like to get started on your custom CBD labels visit our InstaProof page. There, you can upload and approve your proof right away! Or if you’d like to speak to a representative, dial (800) 750-7764. LabelValue is open Monday through Friday.

 Save 10% off your first LabelValue order |
Save 10% off your first LabelValue order |